This article was released as Pharm Edaily Premium Content on March 24, 2026, at 8:00 AM.
[Jiwan Kim, Edaily Reporter] On March 23, Korea’s pharmaceutical and biotech sector showed selective strength despite a sharp decline in both the KOSPI and KOSDAQ markets, with only companies backed by clear catalysts posting gains.
Cha Vaccine Institute surged to the daily limit as its business restructuring strategy following Solux’s acquisition became clearer. Kyongbo Pharmaceutical climbed on expectations of enhanced global manufacturing credibility after passing a U.S. FDA inspection. TiumBio also gained momentum as its immuno oncology candidate TU2218 demonstrated a 70% objective response rate (ORR) in head and neck cancer trials, fueling expectations for a potential global licensing deal.
These stock movements were driven by factors such as governance changes, prospects for U.S. market entry and strong clinical outcomes.
 |
Cha Vaccine Institute CI. (Phot=Cha Vaccine Institute) |
<이미지를 클릭하시면 크게 보실 수 있습니다> |
Cha Vaccine gains on Solux deal
Cha Vaccine Institute saw its share price soar after the market reacted positively to the detailed business strategy unveiled following Solux’s acquisition of management control.
According to KG Zeroin’s MP Doctor data, the stock jumped 29.90% (1,220 won) to close at 5,300 won, hitting the daily upper limit. Investors viewed the move not as a simple change in ownership but as the materialization of a long-term growth strategy.
Solux clarified that the acquisition was a strategic decision aimed at strengthening its existing lighting business while securing a new growth engine by linking Cha Vaccine Institute with AriBio.
The company plans to establish a dual-structure business model maintaining stable earnings from its lighting segment while expanding long-term growth through its biotech division. This reflects a balanced portfolio strategy combining cash-generating businesses with high growth new ventures.
Each affiliate’s role is also becoming clearer. AriBio will focus on the clinical development and commercialization of its oral Alzheimer’s treatment AR1001, while Cha Vaccine Institute will strengthen its R&D capabilities in vaccines, immunology, and NK cell-based therapies.
Solux aims to act as a platform connecting these entities, building a structure in which both lighting and biotech businesses grow simultaneously. The strategy is designed to reduce dependency on specific pipelines while securing diversified growth drivers.
The company also highlighted that CHA Biotech remains the second-largest shareholder after the deal, interpreting this as a signal of confidence in future business expansion and corporate value.
A Solux official said, “This acquisition of Cha Vaccine Institute represents a process of adding a new growth axis on top of our existing business foundation,” adding, “We will advance our lighting business while gradually materializing a growth structure that connects Cha Vaccine Institute and AriBio.”
Kyongbo Pharma rises on FDA inspection clearance
Kyongbo Pharmaceutical continued its upward momentum following positive sentiment from its FDA inspection clearance at its Asan plant. The stock rose 810 won (11.33%) to close at 7,960 won.
Investor sentiment was boosted by news that the Asan facility had passed an on-site inspection by the U.S. Food and Drug Administration (FDA). The easing of regulatory risks surrounding production facilities raised expectations that the company’s API and contract manufacturing (CMO) businesses could gain greater global credibility.
On March 18, Kyongbo Pharmaceutical announced that it had received a Voluntary Action Indicated (VAI) classification following the FDA inspection.
FDA inspection outcomes are typically categorized into three levels: No Action Indicated (NAI), Voluntary Action Indicated (VAI), and Official Action Indicated (OAI).
A VAI classification indicates that while minor issues were identified, they do not significantly impact product quality or safety. No mandatory regulatory actions are required, and companies can address the findings voluntarily meaning there are no substantial restrictions on production or supply.
In contrast, NAI represents the highest rating with no observations, while OAI indicates significant violations that could lead to enforcement actions such as import alerts or warning letters.
The inspection at the Asan plant covered facilities producing ceftobiprole, a fifth generation cephalosporin antibiotic and lenalidomide, an immunomodulatory anticancer drug intended for the U.S. market.
The FDA reviewed overall compliance with current Good Manufacturing Practice (cGMP), including manufacturing processes, system design, quality control, and raw material management. Although some improvements were recommended, they were not deemed critical to product quality.
Industry observers view the VAI classification as confirmation that the facility largely meets global cGMP standards, potentially boosting confidence among overseas clients and supporting future order growth.
A company official stated, “The VAI classification indicates that our Asan plant has an overall management system that meets cGMP standards,” adding, “We will continue to enhance our production environment and quality through proactive investments to strengthen our global competitiveness.”
 |
Kyongbo Pharmaceutical headquarters. (Photo=Kyongbo Pharmaceutical) |
<이미지를 클릭하시면 크게 보실 수 있습니다> |
TiumBio gains on ‘70% ORR’ in head and neck cancer
TiumBio drew strong market attention after its oral immuno oncology candidate TU2218 achieved a 70% objective response rate (ORR) in a Phase 2a trial in combination with Keytruda for head and neck cancer.
The result significantly exceeds the response rates typically observed with PD-1 inhibitors, leading to rising expectations for a global licensing deal. Earlier, Pharma EDaily highlighted this milestone in a premium article titled “ORR 70% shakes the head and neck cancer landscape… TiumBio’s TU2218 heads toward a global deal.”
The data showed partial responses in 12 out of 17 patients, translating into an ORR of 70.6% and a disease control rate (DCR) of 82.4%.
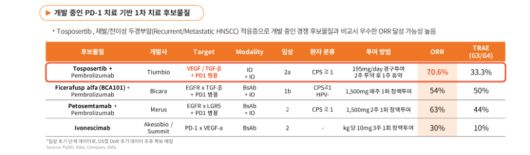 |
TiumBio’s TU2218 (Tosposertib) recorded the highest objective response rate (ORR) in a PD-1–based head and neck cancer trial. (Source=TiumBio) |
<이미지를 클릭하시면 크게 보실 수 있습니다> |
This compares favorably with competing therapies in the same indication, which generally report ORRs in the range of 30–60%. The results also demonstrate nearly double the efficacy compared to Keytruda monotherapy or chemotherapy combinations, underscoring their clinical significance.
TU2218 differentiates itself by not only activating immune responses but also reshaping the tumor microenvironment. By blocking TGF-β signaling, it removes barriers that prevent immune cell infiltration, while inhibiting VEGFR2 suppresses tumor angiogenesis—thereby enhancing the efficacy of immunotherapy.
The market responded immediately with TiumBio shares rising 8.24% (830 won) to close at 10,900 won. Industry experts view TU2218 as a leading candidate targeting the tumor microenvironment and are closely watching its potential for global partnerships.
A TiumBio official said, “The data secured so far already demonstrates strong competitiveness,” adding, “We aim to deliver topline results from the Phase 2a trial within this year and actively pursue global partnering opportunities.”
이 기사의 카테고리는 언론사의 분류를 따릅니다.
기사가 속한 카테고리는 언론사가 분류합니다.
언론사는 한 기사를 두 개 이상의 카테고리로 분류할 수 있습니다.
언론사는 한 기사를 두 개 이상의 카테고리로 분류할 수 있습니다.


